Saturday, May 3, 2014
Periodic table
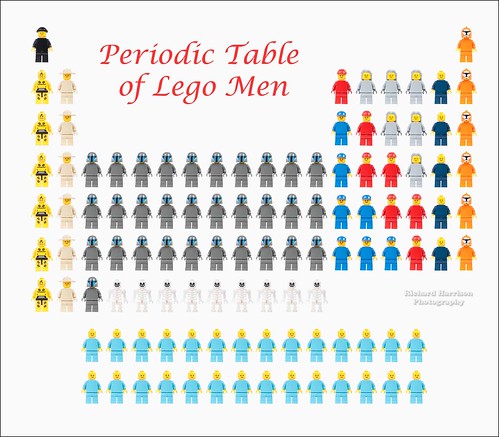
The periodic table, here by Richard Harrison, lays out all of the different elements, the different types of atoms that make up the world around us. There are a little over a hundred different types known, and some scientists are continuing to work on finding bigger and bigger elements. The elements get bigger and bigger as you read left to right and down the table (technically this is increasing atomic number). The key insight that helped Mendeleev categorize the elements was that when you list them in order there are repeating, or periodic properties. The lines are broken up so that everything within a column has these periodic properties. Chemists can look at where an element is on the table, and what other elements are nearby, and make conclusions about the properties of that element.


Labels:
chemistry
Subscribe to:
Post Comments (Atom)
No comments:
Post a Comment